-
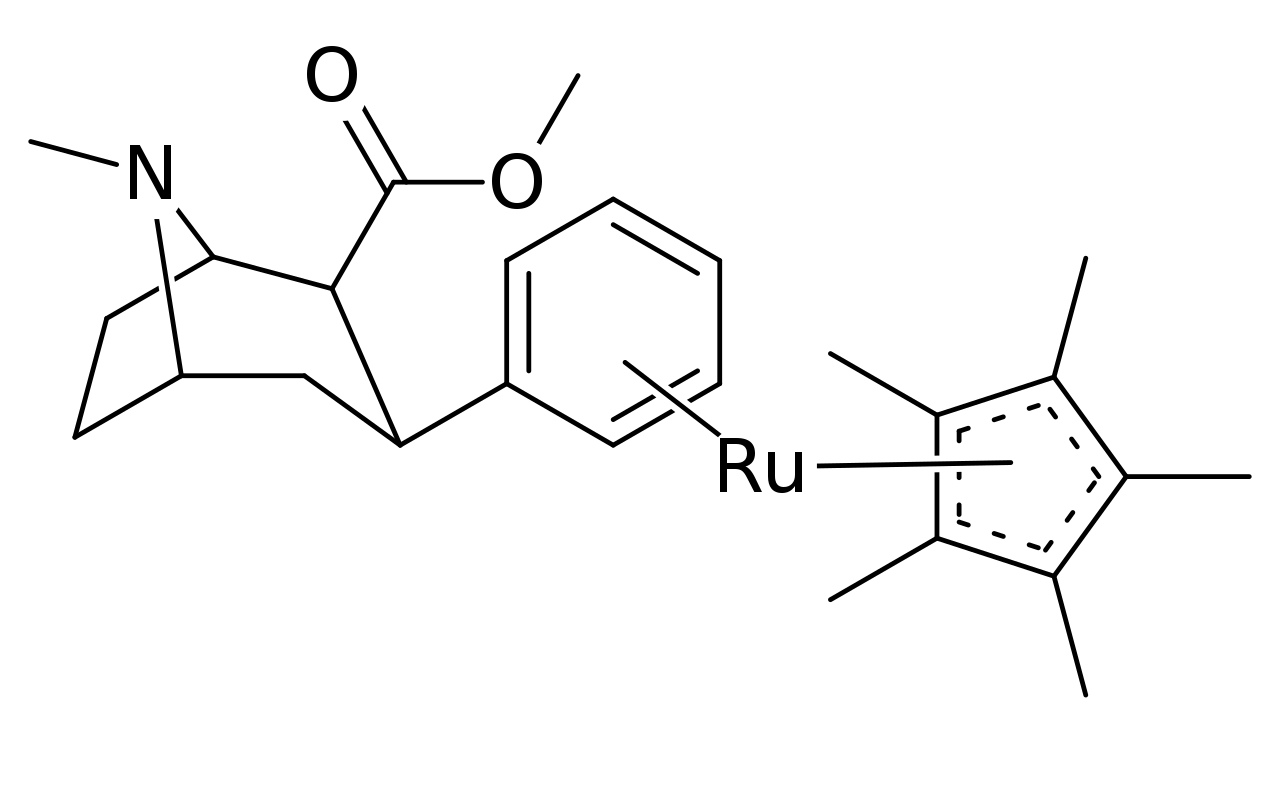
Addition in pharmacological active compounds
Several variants of pi coordinated phenyls have even been tested using transition metals for stacking η6-phenyltropanes, using cyclopentadienyl and tricarbonyl in place of a benzene. Which in the case of the tricarbonyl doubled the compound’s affinity for its intended ligand site (posited as due to resultant electrostatic influences being more conducive to the target).
-
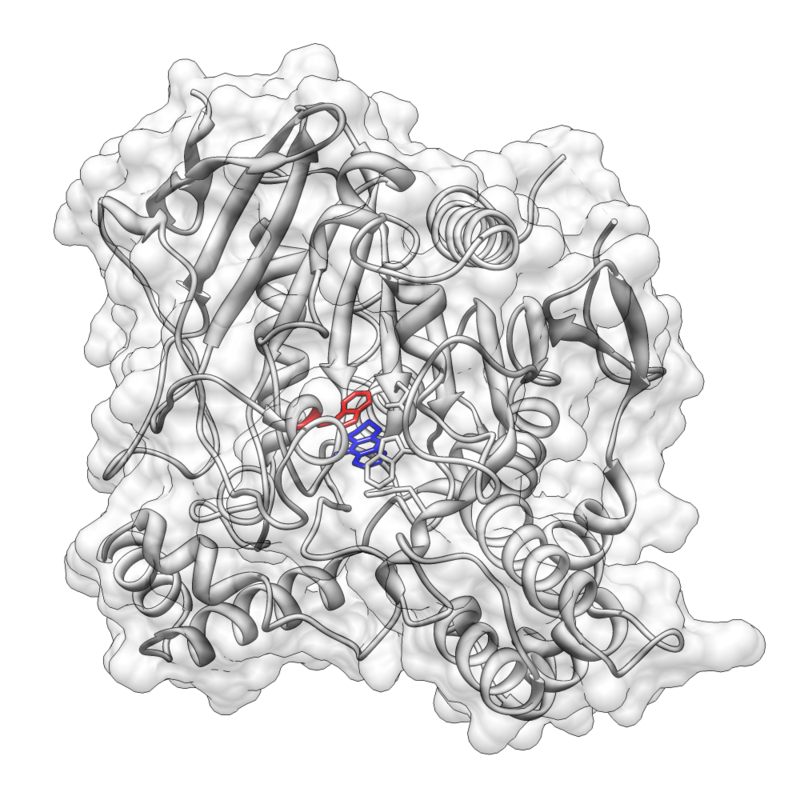
Pi stacking is prevalent in protein crystal structures and also contributes to the interactions between small molecules and proteins
As a result, pi–pi and cation–pi interactions are important factors in rational drug design. One example is the FDA-approved acetylcholinesterase (AChE) inhibitor tacrine which is used in the treatment of Alzheimer’s disease. Tacrine is proposed to have a pi stacking interaction with the indolic ring of Trp84, and this interaction has been exploited in the rational design of novel AChE
-
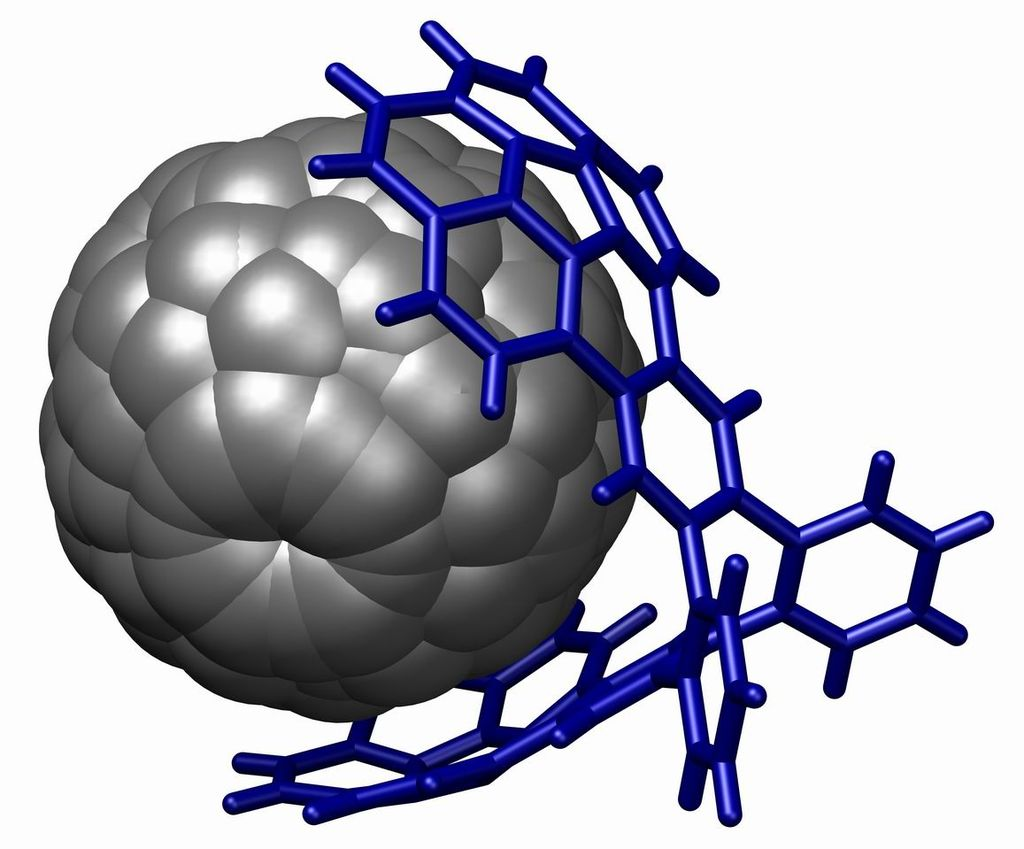
A powerful demonstration of stacking is found in the buckycatcher
This molecular tweezer is based on two concave buckybowls with a perfect fit for one convex fullerene molecule. Complexation takes place simply by evaporating a toluene solution containing both compounds. In solution an association constant of 8600 M−1 is measured based on changes in NMR chemical shifts.
-
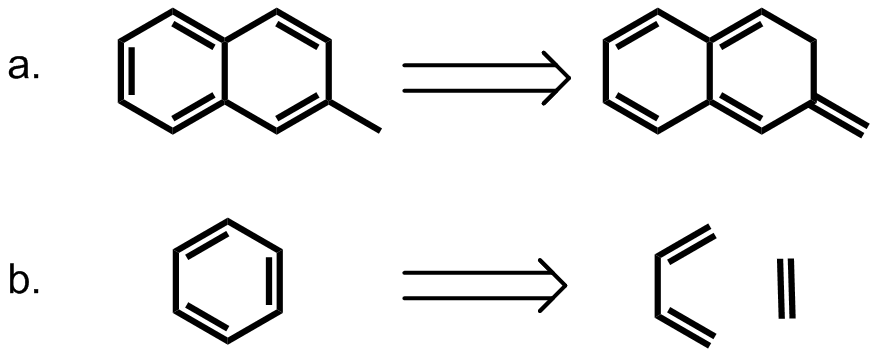
Requirement of aromaticity
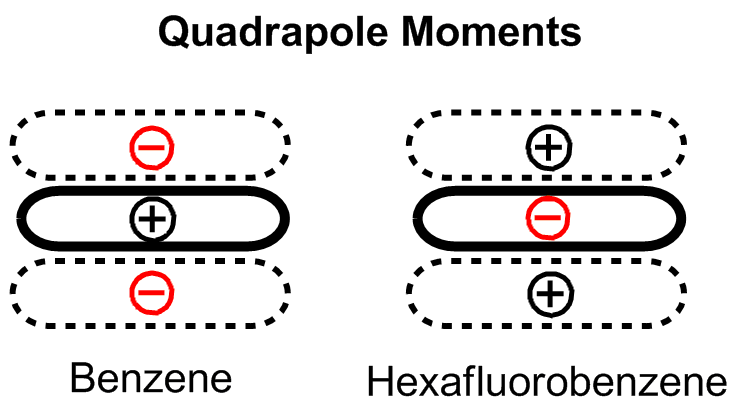
The conventional understanding of pi stacking involves quadrupole interactions between delocalized electrons in p-orbitals. In other words, aromaticity should be required for this interaction to occur. However, several groups have provided contrary evidence, calling into question whether pi stacking is a unique phenomenon or whether it extends to other neutral, closed-shell molecules. In an experiment
-
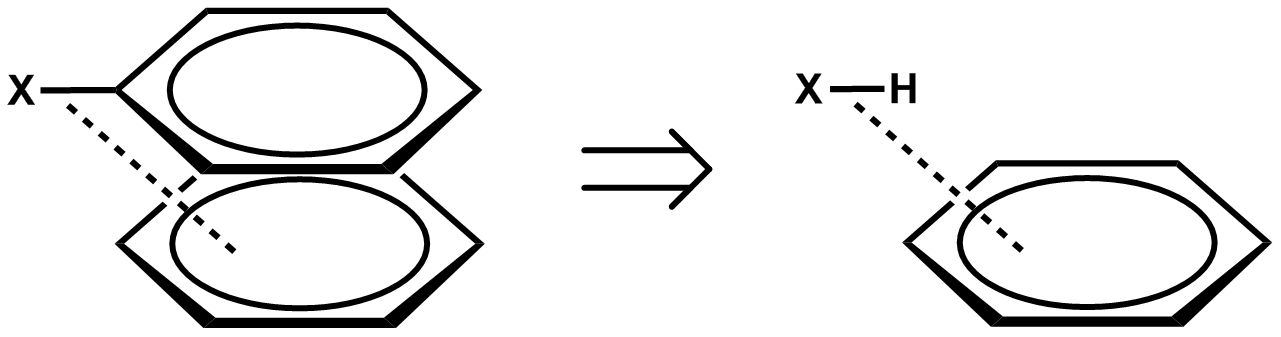
Direct interaction model (substituent effects)
The Hunter–Sanders model has been criticized by numerous research groups offering contradictory experimental and computational evidence of pi stacking interactions that are not governed primarily by electrostatic effects. The clearest experimental evidence against electrostatic substituent effects was reported by Rashkin and Waters. They used meta- and para-substituted N-benzyl-2-(2-fluorophenyl)-pyridinium bromides, which stack in a parallel displaced
-
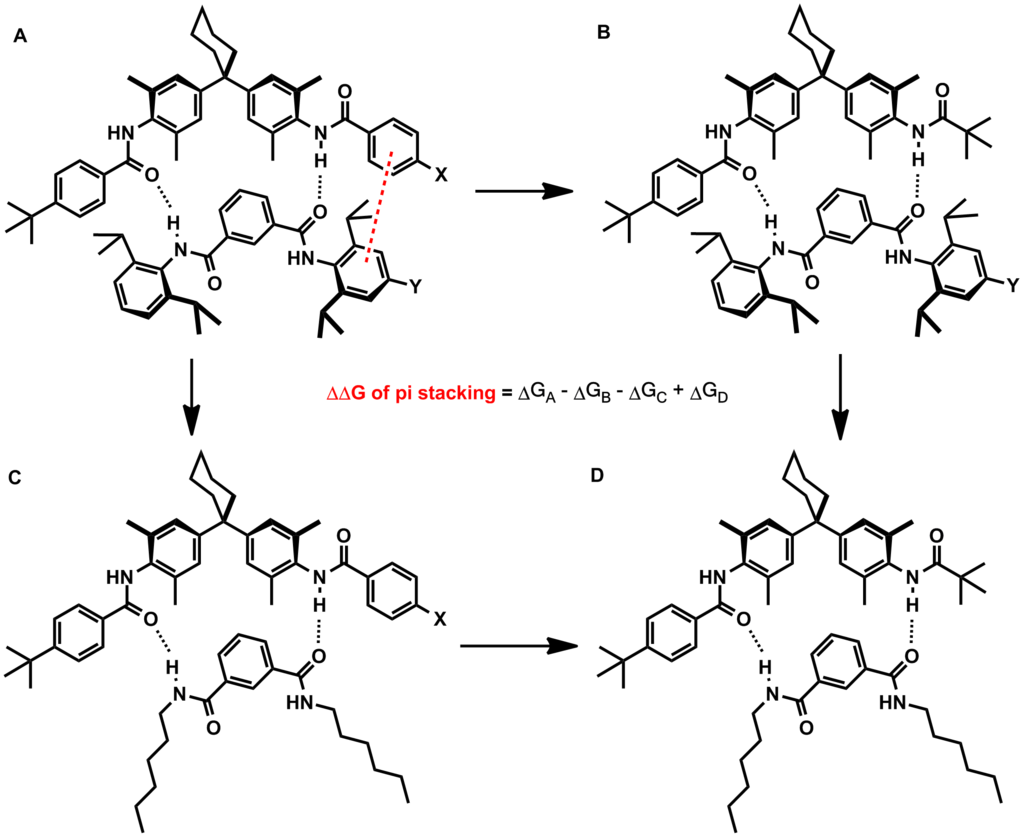
Electrostatic model (substituent effects)
An early model for the role of substituents in pi stacking interactions was proposed by Hunter and Sanders. They used a simple mathematical model based on sigma and pi atomic charges, relative orientations, and van der Waals interactions to qualitatively determine that electrostatics are dominant in substituent effects. According to their model, electron-withdrawing groups reduce the negative
-

Substituent effects
The ability to fine-tune pi stacking interactions would be useful in numerous synthetic efforts. One example would be to increase the binding affinity of a small-molecule inhibitor to an enzyme pocket containing aromatic residues. The effects of heteroatoms and substituents on pi stacking interactions is difficult to model and a matter of debate.
-
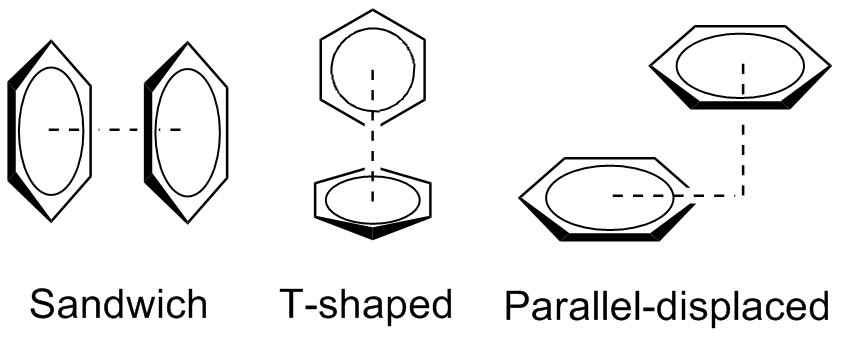
Geometric configurations
The preferred geometries of the benzene dimer have been modeled at a high level of theory with MP2-R12/A computations and very large counterpoise-corrected aug-cc-PVTZ basis sets. The two most stable conformations are the parallel displaced and T-shaped, which are essentially isoenergetic. In contrast, the sandwich configuration maximizes overlap of the pi system, which destabilizes the
-

Evidence against pi stacking
The benzene dimer is the prototypical system for the study of pi stacking, and is experimentally bound by 8–12 kJ/mol (2–3 kcal/mol) in the gas phase with a separation of 4.96 Å between the centers of mass for the T-shaped dimer. The small binding energy makes the benzene dimer difficult to study experimentally, and the dimer itself
-
Pi stacking aka π–π stacking
In chemistry, pi stacking (also called π–π stacking) refers to the presumptive attractive, noncovalent pi interactions (orbital overlap) between the pi bonds of aromatic rings. However this is a misleading description of the phenomena since direct stacking of aromatic rings (the “sandwich interaction”) is electrostatically repulsive. What is more commonly observed (see figure below) is either a staggered stacking (parallel displaced) or pi-teeing (perpendicular T-shaped) interaction both of which are electrostatic attractive For
Recent Posts
- The Titles of Nobility Clauses Are Screaming and Nobody Is Listening

- 🛡️ 50 Corporate TON Violations (and there are many more)

- TON makes ERA redundant because it forbids legal caste creation at the root

- Aristocracy is a poisonous weed and the TON clauses are weedkiller (in the unhinged spirit of Justice Scalia)

- Adams and Jefferson on Aristocracy: A Structural Disagreement in the Early Republic

Tags
2025 agriculture alchemy anthropology archaeology art biochemistry biography biology chemistry civil rights crystallography culture duplicate ecology economics endocrinology entertainment entomology folklore genetics geology glossary hematology history immunology law linguistics materials science medicine mythology neurology philosophy physiology politics psychology public health religion reproduction science smoker's paradox symbolism systems theory technology toxicology